Fig. 1:
Scheme of refined model for bacterial biofilm transfer on human epithelial lung cells. The biofilm are unlike before bioprinted on the cells.
To fight lung infections, medical science explores disease progression and develops new anti-infectives and therapeutic approaches. Biofilm-forming bacteria present a major challenge for medicine. For patients with cystic fibrosis, these bacteria are a special threat, because they can colonize the thick and viscous mucus of these persons.
In the course of this research, animal experiments are still the standard model, despite of several known drawbacks. Usually, the “animal trial objects“ (rats, mouse, cats, cavies, pigs, monkeys) are infected with bacteria-loaded agar-beads via the intratracheal or intranasal route. However, such animal models are not just ethical but also scientifically questionable.
Advanced and in vivo like in vitro models could not just offer a simple substitute, but be an even better alternative to receive preclinical drug efficacy data of new anti-infectives. One major problem is however, that bacterial biofilms cannot be grown directly on human cells, because their rapid growth quickly kills the cells. In a new approach the bacterial biofilms are grown separately and transferred onto the cells. Still, with this method the biofilm can be transferred neither in a defined nor in a reproducible way.
Therefore, this project aims to bioprint bacterial biofilms in a hydrogel on human lung epithelial cells in a reproducible and defined manner (figure 1). Subsequently, the model is supposed to be used as a platform for the investigation of new anti-infectives.
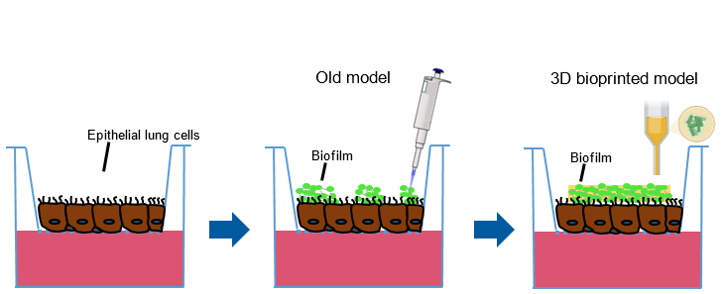
Fig. 1:
Scheme of refined model for bacterial biofilm transfer on human epithelial lung cells. The biofilm are unlike before bioprinted on the cells.
Pseudomonas aeruginosa bacteria (e.g. PA14) will be grown in a printable hydrogel to form biofilm material. The biofilms will then be printed onto human lung epithelial cells. Afterwards, the model will be characterized with special regards to cell viability and barrier properties as well as cytokine release. The application of the model via treatment with suitable anti-infectives (e.g. Tobramycin, Colistin or QS-inhibitors) shall be demonstrated by observing changing barrier properties and cytokine releases.
Research group Prof. Dr. Claus-Michael Lehr
"Drug Delivery Across Biological Barriers"
Helmholtz-Institute for Pharmaceutical Research Saarland
Campus E8 1, 66123 Saarbrücken
07/2021 - 01/2024
[Translate to English:] Zurück zur Projektliste