Background
Botulinum toxin is a bacterial exotoxin. It is produced by the anaerobic spore-forming bacterium Clostridium botulinum and is one of the most potent bacterial toxins known. The lethal dose for humans is about 1 ng / kg body weight. Botulinum toxin is a neurotoxin that inhibits the release of neurotransmitters from the nerve endings, which stimulate muscle contraction. Thus it leads to flaccid paralysis of the affected muscles. Death occurs from paralysis of the respiratory muscles.
Botulinum toxin is a protein that consists of two subunits. The large subunit ensures that the small subunit can penetrate into nerve cells. In the nerve cell, the small subunit destroys proteins that are necessary for the release of the transmitter substances from the nerve cells. There are several serotypes of botulinum toxin. Each serotype uses different cellular proteins to bind to the nerve cells and the small subunits of the distinct serotypes cleave unique proteins required for neurotransmitter release.
The bacteria thrive in poorly sterilized food stored in air tight containers, such as improperly processed canned food. Consumption of food contaminated with these bacteria leads to the often fatal disease of botulism. In low doses and administered locally, botulinum toxin can be used as a drug to treat illnesses that result from a sustained contraction (spasm) of a single muscle or a group of muscles such as the blepharospasm or the spastic torticollis. The most frequent therapeutic use of botulinum toxin is, however, in cosmetic medicine, where it is used to suppress the formation of wrinkles by paralyzing the subcutaneous skin muscles causing the wrinkles. In this case, the botulinum toxin is injected at a low concentration in the corresponding regions of the skin.
For the therapeutic application, Botulinum toxin is obtained from cultures of Clostridia. Due to the necessary purification steps, part of the protein is inactivated. Therefore, the final preparation consists of varying proportions of functional and inactive proteins. Because of the high toxicity, it is extremely important to accurately determine the activity of each preparation. It is mandatory to assess the activity of both the large and the small subunit. Currently, the most reliable method to achieve this is the determination of the biological effect in the whole animal. To this end, the concentration of the preparation is measured, at which half of the treated mice die of respiratory paralysis. For this test, about 300,000 mice annually worldwide suffer an agonizing death caused by suffocation. (http://altweb.jhsph.edu/sebin/c/g/altex_2_10_bitz.pdf; accessed 14. 6. 2012).
Project:
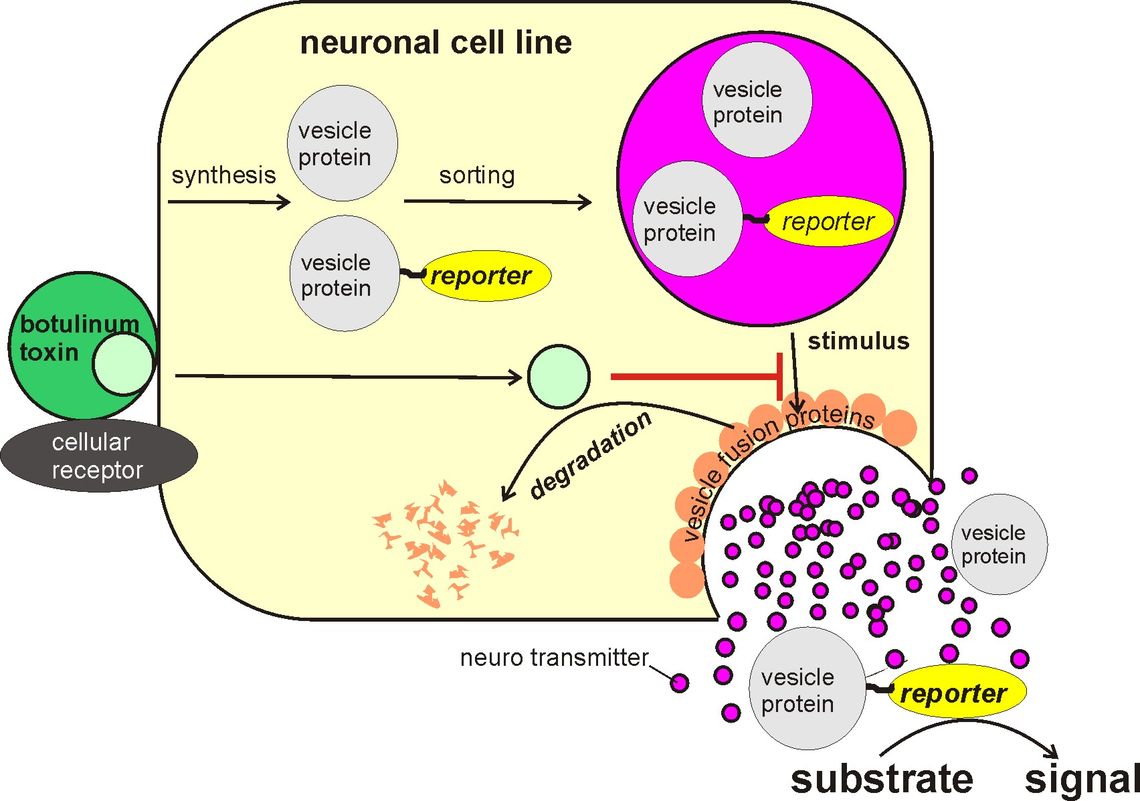
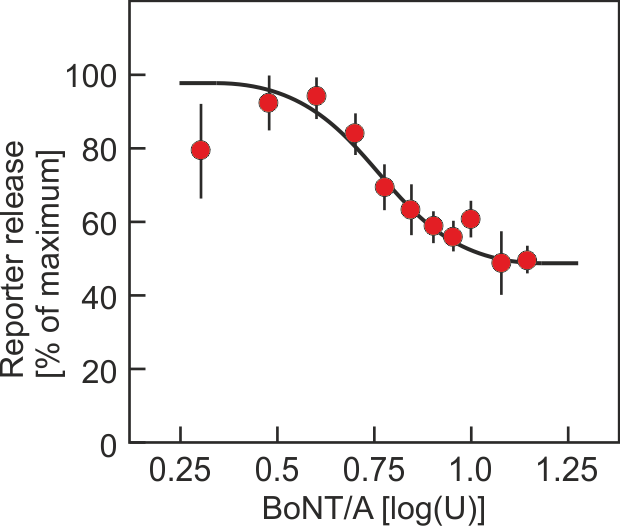
In this project, a cell-based assay for the determination of the activity of botulinum toxin was established. It is suitable to largely replace the animal experiment. The newly established assay differs from all in vitro tests developed so far: The assay determines the end point of the Botulinum toxin action, i. e. the inhibition of the neurotransmitter release from the nerve ending by the detection of a reporter that is released concomitantly with the neurotransmitter. This warrants that the activity of both the large and the small subunit of the toxin are assessed by the assay. In theory, the assay should be applicable to all serotypes of botulinum toxin.
The principle (Fig. 1) of the test is based on the fact that proteins are directed towards particular subcellular compartments of the cell by certain amino acid sequences (address sequences). Using appropriate amino acid sequences, proteins can be directed into the storage vesicles from which neurotransmitters are released. These proteins will be released together with the neurotransmitters and their release will be inhibited, if the transmitter release is inhibited. If an enzyme is chosen as reporter that is not naturally produced by the cells, the release of the reporter can be determined by measuring the enzyme activity in the cell culture supernatant after stimulation of the cell. The stimulus-dependent release of reporter enzyme decreases if the neurotransmitter release is inhibited by Botulinum toxin.


 English
English